Nirmatrelvir Ritonavir Tablets IP
Price 3000 INR/ Pack
Nirmatrelvir Ritonavir Tablets IP Specification
- Pacakaging (Quantity Per Box)
- 30 tablets (20 Nirmatrelvir & 10 Ritonavir)
- Origin of Medicine
- India
- Salt Composition
- Nirmatrelvir and Ritonavir
- Indication
- Treatment of COVID-19
- Life Span
- 24 months from manufacturing date
- Brand Name
- Nirmatrelvir Ritonavir Tablets IP
- Dosage Form
- Oral tablet
- Packaging Type
- Blister packaging
- Drug Type
- Antiviral prescription drug
- Ingredients
- Nirmatrelvir 150 mg and Ritonavir 100 mg
- Physical Form
- Other
- Function
- Other
- Recommended For
- Adults with mild to moderate COVID-19 at risk of developing severe disease
- Dosage
- As directed by physician, typically 2 tablets of Nirmatrelvir and 1 tablet of Ritonavir together twice daily for 5 days
- Dosage Guidelines
- Swallow tablets whole, do not crush or split
- Suitable For
- Adults
- Quantity
- Co-packaged tablet strip
- Storage Instructions
- Store below 30C, protect from moisture and light
- Clinical Use
- Emergency use authorized for high risk COVID-19 patients
- Tablet Shape
- Oval
- Administration Route
- Oral
- Contraindications
- Not recommended in patients with severe renal or hepatic impairment
- Manufacturing Standard
- IP (Indian Pharmacopoeia)
- Marketed By
- Certified Indian pharmaceutical manufacturers
- Colour of Tablet
- Nirmatrelvir - pink, Ritonavir - white
- Packing Material
- Aluminum-Aluminum blister strips
- Prescription Required
- Yes
- Interaction Warning
- May interact with other medications metabolized by CYP3A
- Side Effects
- Possible nausea, diarrhea, altered taste
- Regulatory Approval
- DCGI approved
Nirmatrelvir Ritonavir Tablets IP Trade Information
- Minimum Order Quantity
- 100 Packs
- Supply Ability
- 5000 Packs Per Month
- Delivery Time
- 10 Days
- Main Domestic Market
- All India
About Nirmatrelvir Ritonavir Tablets IP
Advanced Application, Features & Competitive Edge
Nirmatrelvir Ritonavir Tablets IP are tailored exclusively for oral administration, applying their potent antiviral action directly where needed. Noteworthy extra features include their IP manufacturing standard, distinct color coding, and sturdy blister packaging for pristine preservation. What sets them apart is their competitive advantage of rapid deployment for high-risk COVID-19 patients, DCGI regulatory approval, and strict adherence to safety protocols-making them a leading choice among global suppliers, exporters, and traders.
Swift Delivery, Secure Packaging & Port Details
Experience quick dispatch and competitive rates when you order Nirmatrelvir Ritonavir Tablets IP. Each offer includes a co-packaged strip of 30 tablets, packed in secure aluminum blister packaging to ensure integrity during transit. Delivery timelines are streamlined, with export options via major Indian FOB ports, so you receive your order promptly and in pristine condition. Trust our efficient supply process for hassle-free fulfillment from India to your location.
FAQ's of Nirmatrelvir Ritonavir Tablets IP:
Q: How should Nirmatrelvir Ritonavir Tablets IP be administered?
A: Nirmatrelvir Ritonavir Tablets IP are taken orally, with the recommended usage being two tablets of Nirmatrelvir and one tablet of Ritonavir taken together, twice daily for five days, as directed by a physician. Tablets should be swallowed whole, not crushed or split.Q: What patients are recommended to use Nirmatrelvir Ritonavir Tablets IP?
A: These tablets are recommended for adults with mild to moderate COVID-19 who are at risk of developing severe disease. They are not advised for individuals with severe renal or hepatic impairment.Q: When is a prescription required for ordering these tablets?
A: A prescription is mandatory for purchasing Nirmatrelvir Ritonavir Tablets IP due to their clinical use and regulatory status. Please consult a healthcare professional for assessment and authorization.Q: Where are Nirmatrelvir Ritonavir Tablets IP manufactured and marketed?
A: These tablets are manufactured and marketed by certified pharmaceutical companies in India, adhering to Indian Pharmacopoeia standards and DCGI regulations.Q: What competitive advantages do Nirmatrelvir Ritonavir Tablets IP offer?
A: Their advantages include emergency use authorization, invincible IP standard manufacturing, reliable DCGI approval, robust packaging, and prompt availability for high-risk COVID-19 patients, making them a preferred choice for exporters and healthcare providers globally.Q: How are the tablets packaged to ensure safety and efficacy?
A: Each co-packaged strip contains both Nirmatrelvir and Ritonavir tablets, sealed in aluminum-aluminum blister strips to preserve their pristine quality, protected from moisture and light, with a lifespan of 24 months.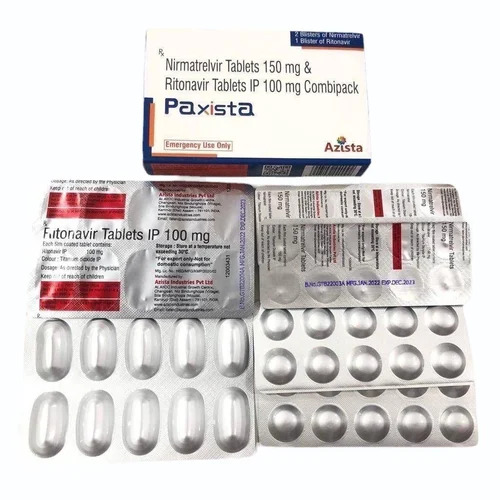

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Pharmaceutical Tablets Category
200mg Favipiravir Tablet
Price 350 INR / Pack
Minimum Order Quantity : 100 Packs
Indication : Treatment of mild to moderate COVID19
Drug Type : Generic Drugs
Suitable For : Adults
Life Span : 24 months
400mg Favipiravir Tablet
Price 150 INR / Pack
Minimum Order Quantity : 100 Packs
Indication : Treatment of influenza and COVID19
Drug Type : Other, Allopathic
Suitable For : Adults
Life Span : 24 months from date of manufacture
Favipiravir Favivent Tablets
Price 400 INR / Pack
Minimum Order Quantity : 100 Packs
Indication : Treatment of mild to moderate COVID19, Influenza
Drug Type : General Medicines
Suitable For : Adults
Life Span : 24 months
Favipiravir 400 Mg Favivir Tablet
Price 200 INR / Pack
Minimum Order Quantity : 100 Packs
Indication : Treatment of COVID19 infection
Drug Type : Other, Allopathic
Suitable For : Adults
Life Span : 24 months

 Send Inquiry
Send Inquiry

 Send Inquiry
Send Inquiry English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese